Eucheuma cottonii as a Natural Source for Type-2 Diabetes Mellitus Management: A Narrative review
DOI:
https://doi.org/10.36733/medicamento.v12i1.12689Keywords:
antidiabetic, anti-inflammatory, antioxidant, Eucheuma cottonii, prebioticAbstract
Background: Type 2 diabetes mellitus is a chronic metabolic disorder with a steadily increasing prevalence worldwide, including in Indonesia. Standard therapeutic approaches for T2DM are commonly associated with adverse effects, which has led to increasing interest in the use of herbal products and nutraceuticals. Eucheuma cottonii, an abundant red seaweed species in Indonesian waters, holds considerable potential as a preventive and therapeutic agent for T2DM due to its bioactive compound content.
Objective: This study aimed to evaluate the potential of E. cottonii as a therapeutic agent through a literature review.
Methods: This study is a literature review. Relevant literature was collected from several databases using specific keywords, and a total of 30 articles that met the inclusion criteria were selected for analysis.
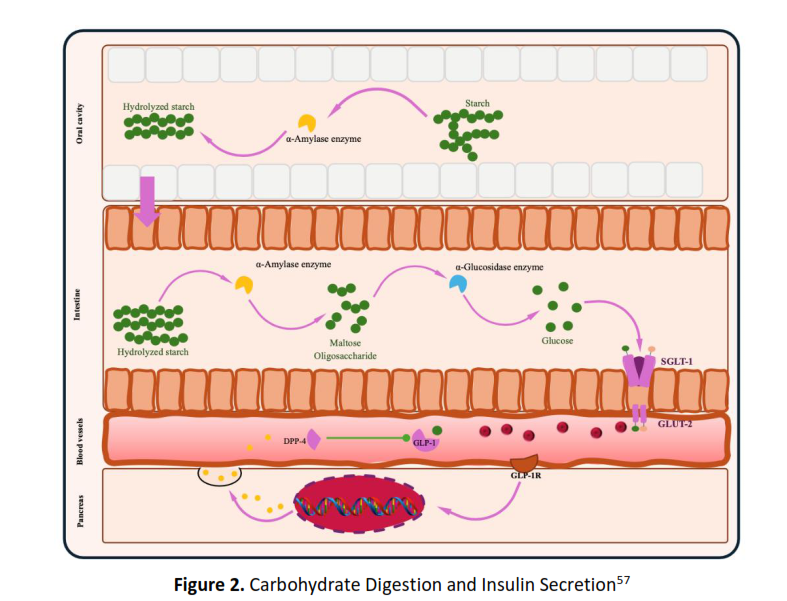
Results: Evidence suggested that E. cottonii comprises numerous bioactive substances compounds including flavonoids, phenolics, sulfated polysaccharides, and carrageenan, which demonstrate antidiabetic activity through multiple mechanisms. These compounds inhibit α-amylase and α-glucosidase enzymes, lower blood glucose levels, and enhance insulin secretion via dipeptidyl peptidase-4 inhibition. The antioxidant activity of E. cottonii is demonstrated by elevated levels of endogenous enzymes (SOD, GPx, GSH) and reduced ROS and MDA, while its anti-inflammatory effects were reflected in decreased expression of pro-inflammatory cytokines and inflammatory enzymes (COX-2, LOX-5). Moreover, E. cottonii functions as a prebiotic, modulating gut microbiota, enhancing the population of Bifidobacterium spp., and improving the Firmicutes/Bacteroidetes ratio, thereby contributing to better glucose metabolism.
Conclusion: Based on these bioactivities, E. cottonii demonstrates strong potential for development as a multifunctional nutraceutical for the prevention and management of T2DM through antihyperglycemic, antioxidant, anti-inflammatory, and gut microbiota–modulating mechanisms.
References
1. Wahidin M, Achadi A, Besral B, Kosen S, Nadjib M, Nurwahyuni A, Ronoatmodjo S, Rahajeng E, Pane M, Kusuma D. Projection of diabetes morbidity and mortality till 2045 in Indonesia based on risk factors and NCD prevention and control programs. Sci Rep. 2024;14(1):5424. doi:10.1038/s41598-024-54563-2
2. Banday MZ, Sameer AS, Nissar S. Pathophysiology of diabetes: An overview. Avicenna J Med. 2020;10(04):174-188. doi:10.4103/ajm.ajm_53_20
3. Kementerian Kesehatan Republik Indonesia. Laporan Hasil Riset Kesehatan Dasar Nasional (Riskesdas) 2018. Badan Peneliti dan Pengembangan Kesehatan; 2019.
4. Kementerian Kesehatan Republik Indonesia. Laporan Hasil Riset Kesehatan Dasar Nasional (Riskesdas) 2007. Badan Penelitian dan Pengembangan Kesehatan; 2008.
5. DeFronzo RA. Pathogenesis of type 2 diabetes mellitus. Medical Clinics of North America. 2004;88(4):787-835. doi:10.1016/j.mcna.2004.04.013
6. Muoio DM, Newgard CB. Molecular and metabolic mechanisms of insulin resistance and β-cell failure in type 2 diabetes. Nat Rev Mol Cell Biol. 2008;9(3):193-205. doi:10.1038/nrm2327
7. Leahy JL. Pathogenesis of Type 2 Diabetes Mellitus. Arch Med Res. 2005;36(3):197-209. doi:10.1016/j.arcmed.2005.01.003
8. Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14(2):88-98. doi:10.1038/nrendo.2017.151
9. Reed J, Bain S, Kanamarlapudi V. A Review of Current Trends with Type 2 Diabetes Epidemiology, Aetiology, Pathogenesis, Treatments and Future Perspectives. Diabetes Metab Syndr Obes. 2021;Volume 14:3567-3602. doi:10.2147/DMSO.S319895
10. Galicia-Garcia U, Benito-Vicente A, Jebari S, Larrea-Sebal A, Siddiqi H, Uribe KB, Ostolaza H, Martín C. Pathophysiology of Type 2 Diabetes Mellitus. Int J Mol Sci. 2020;21(17):6275. doi:10.3390/ijms21176275
11. Weinberg Sibony R, Segev O, Dor S, Raz I. Overview of oxidative stress and inflammation in diabetes. J Diabetes. 2024;16(10):e70014. doi:10.1111/1753-0407.70014
12. Derosa G, Maffioli P. Mini-Special Issue paper Management of diabetic patients with hypoglycemic agents α-Glucosidase inhibitors and their use in clinical practice. Archives of Medical Science. 2012;5:899-906. doi:10.5114/aoms.2012.31621
13. Lankatillake C, Huynh T, Dias DA. Understanding glycaemic control and current approaches for screening antidiabetic natural products from evidence-based medicinal plants. Plant Methods. 2019;15(1):105. doi:10.1186/s13007-019-0487-8
14. Appuhamy JADRN, Kebreab E, Simon M, Yada R, Milligan LP, France J. Effects of diet and exercise interventions on diabetes risk factors in adults without diabetes: meta-analyses of controlled trials. Diabetol Metab Syndr. 2014;6(1):127. doi:10.1186/1758-5996-6-127
15. Le Y, Wang B, Xue M. Nutraceuticals use and type 2 diabetes mellitus. Curr Opin Pharmacol. 2022;62:168-176. doi:10.1016/j.coph.2021.12.004
16. Wanyonyi S, Du Preez R, Brown L, Paul N, Panchal S. Kappaphycus alvarezii as a Food Supplement Prevents Diet-Induced Metabolic Syndrome in Rats. Nutrients. 2017;9(11):1261. doi:10.3390/nu9111261
17. Basyuni M, Puspita M, Rahmania R, Albasri H, Pratama I, Purbani D, Aznawi AA, Mubaraq A, Al Mustaniroh SS, Menne F, Rahmila YI, Salmo III SG, Susilowati A, Larekeng SH, Ardli E, Kajita T. Current biodiversity status, distribution, and prospects of seaweed in Indonesia: A systematic review. Heliyon. 2024;10(10):e31073. doi:10.1016/j.heliyon.2024.e31073
18. Nunes A, Azevedo GZ, de Souza Dutra F, dos Santos BR, Schneider AR, Oliveira ER, Moura S, Vianello F, Maraschin M, Lima GPP. Uses and applications of the red seaweed Kappaphycus alvarezii: a systematic review. J Appl Phycol. 2024;36(6):3409-3450. doi:10.1007/s10811-024-03270-6
19. Das D, Arulkumar A, Paramasivam S, Lopez-Santamarina A, Del Carmen Mondragon A, Miranda Lopez JM. Phytochemical Constituents, Antimicrobial Properties and Bioactivity of Marine Red Seaweed (Kappaphycus alvarezii) and Seagrass (Cymodocea serrulata). Foods. 2023;12(14). doi:10.3390/foods12142811
20. Bahrun B, Melanie H, Artanti N, Anita Y, Marraskuranto E, Tuwo A, Miyamae Y. A review of chemical diversity of Kappaphycus alvarezii: biological properties, health benefits, and food applications. Phytochemistry Reviews. Published online November 13, 2025. doi:10.1007/s11101-025-10197-4
21. Shihab S, Jacob A, Mathew J, Ann Mathew A, Shanamitha A. A Review on the Potential Anti-inflammatory Properties of <i>Kappaphycus alvarezii</i> Journal of Natural Remedies. Published online June 13, 2023:361-371. doi:10.18311/jnr/2023/32082
22. Sarian MN, Ahmed QU, Mat So’ad SZ, Alhassan AM, Murugesu S, Perumal V, Syed Mohamad SNA, Khatib A, Latip J. Antioxidant and Antidiabetic Effects of Flavonoids: A Structure-Activity Relationship Based Study. Biomed Res Int. 2017;2017:1-14. doi:10.1155/2017/8386065
23. Shamsudin NF, Ahmed QU, Mahmood S, Shah SAA, Sarian MN, Khattak MMAK, Khatib A, Sabere ASM, Yusoff YM, Latip J. Flavonoids as Antidiabetic and Anti-Inflammatory Agents: A Review on Structural Activity Relationship-Based Studies and Meta-Analysis. Int J Mol Sci. 2022;23(20):12605. doi:10.3390/ijms232012605
24. Brown L, Poudyal H, Panchal SK. Functional foods as potential therapeutic options for metabolic syndrome. Obesity Reviews. 2015;16(11):914-941. doi:10.1111/obr.12313
25. Suganya AM, Sanjivkumar M, Chandran MN, Palavesam A, Immanuel G. Pharmacological importance of sulphated polysaccharide carrageenan from red seaweed Kappaphycus alvarezii in comparison with commercial carrageenan. Biomedicine & Pharmacotherapy. 2016;84:1300-1312. doi:10.1016/j.biopha.2016.10.067
26. Yustisia I, Hardjo M, Hasri, Sari WON, Kasmiati. Optimization and characterization of Kappaphycus alvarezii and κ-carrageenan hydrolysates with potential biomedical applications. Food Hydrocolloids for Health. 2025;7:100205. doi:10.1016/j.fhfh.2025.100205
27. Makkar F, Chakraborty K. Antidiabetic and anti-inflammatory potential of sulphated polygalactans from red seaweeds Kappaphycus alvarezii and Gracilaria opuntia. Int J Food Prop. 2017;20(6):1326-1337. doi:10.1080/10942912.2016.1209216
28. Cyriac B, Eswaran K. Anti- hyperglycemic effect of aqueous extract of Kappaphycus alvarezii (Doty) Doty ex. P. Silva in alloxan-induced diabetic rats. J Appl Phycol. 2016;28(4):2507-2513. doi:10.1007/s10811-015-0762-7
29. Sanjivkumar M, Chandran MN, Suganya AM, Immanuel G. Investigation on bio-properties and in-vivo antioxidant potential of carrageenans against alloxan induced oxidative stress in Wistar albino rats. Int J Biol Macromol. 2020;151:650-662. doi:10.1016/j.ijbiomac.2020.02.227
30. Sun KL, Gao M, Wang YZ, Li XR, Wang P, Wang B. Antioxidant Peptides From Protein Hydrolysate of Marine Red Algae Eucheuma cottonii: Preparation, Identification, and Cytoprotective Mechanisms on H2O2 Oxidative Damaged HUVECs. Front Microbiol. 2022;13. doi:10.3389/fmicb.2022.791248
31. Yulianti E, Sunarti, Wahyuningsih MSH. The effect of Kappaphycus alvarezii fraction on plasma glucose, Advanced Glycation End-products formation, and renal RAGE gene expression. Heliyon. 2021;7(1):e05978. doi:10.1016/j.heliyon.2021.e05978
32. Yulianti E, Sunarti, Wahyuningsih MSH. The effect of Kappaphycus alvarezii active fraction on oxidative stress and inflammation in streptozotocin and nicotinamide-induced diabetic rats. BMC Complement Med Ther. 2022;22(1):15. doi:10.1186/s12906-021-03496-8
33. Vun-Sang S, Sivapragasam G, Yong WTL, Benjamin MAZ, Iqbal M. Assessment of Antioxidant and Hepatoprotective Effects of Kappaphycus alvarezii (Doty) Doty ex Silva Against Carbon Tetrachloride-Induced Liver Injury in Rats. Journal of Tropical Biology & Conservation (JTBC). 2024;21:73-87. doi:10.51200/jtbc.v21i.5420
34. Wardani G, Farida N, Andayani R, Kuntoro M, Sudjarwo S. The potency of red seaweed (Eucheuma cottonii) extracts as hepatoprotector on lead acetate-induced hepatotoxicity in mice. Pharmacognosy Res. 2017;9(3):282. doi:10.4103/pr.pr_69_16
35. Das D, Arulkumar A, Paramasivam S, Lopez-Santamarina A, del Carmen Mondragon A, Miranda Lopez JM. Phytochemical Constituents, Antimicrobial Properties and Bioactivity of Marine Red Seaweed (Kappaphycus alvarezii) and Seagrass (Cymodocea serrulata). Foods. 2023;12(14):2811. doi:10.3390/foods12142811
36. Rudke AR, da Silva M, Andrade CJ de, Vitali L, Ferreira SRS. Green extraction of phenolic compounds and carrageenan from the red alga Kappaphycus alvarezii. Algal Res. 2022;67:102866. doi:10.1016/j.algal.2022.102866
37. Vaghela P, Das AK, Trivedi K, Anand KGV, Shinde P, Ghosh A. Characterization and metabolomics profiling of Kappaphycus alvarezii seaweed extract. Algal Res. 2022;66:102774. doi:10.1016/j.algal.2022.102774
38. Regina LZL, Sharon KTF, Lim LH, Su SF, Teo S Sen. Determination of antioxidant activity, chlorophyll and carotenoids content of Kappaphycus alvarezii: Comparison of seaweed with traditional Chinese Medicines. Res J Biotechnol. 2024;19(4):24-31. doi:10.25303/1904rjbt024031
39. Araújo PG, Nardelli AE, Duran R, Pereira MS, Gelli VC, Mandalka A, Eisner P, Fujii MT, Chow F. Seasonal variation of nutritional and antioxidant properties of different Kappaphycus alvarezii strains (Rhodophyta) farmed in Brazil. J Appl Phycol. 2022;34(3):1677-1691. doi:10.1007/s10811-022-02739-6
40. Teo BSX, Gan RY, Abdul Aziz S, Sirirak T, Mohd Asmani MF, Yusuf E. In vitro evaluation of antioxidant and antibacterial activities of Eucheuma cottonii extract and its in vivo evaluation of the wound‐healing activity in mice. J Cosmet Dermatol. 2021;20(3):993-1001. doi:10.1111/jocd.13624
41. Makkar F, Chakraborty K. Antioxidative sulphated polygalactans from marine macroalgae as angiotensin-I converting enzyme inhibitors. Nat Prod Res. 2018;32(17):2100-2106. doi:10.1080/14786419.2017.1363756
42. Araújo PG, Nardelli AE, Fujii MT, Chow F. Antioxidant properties of different strains of Kappaphycus alvarezii (Rhodophyta) farmed on the Brazilian coast. Phycologia. 2020;59(3):272-279. doi:10.1080/00318884.2020.1736878
43. Papitha R, Selvaraj CI, Palanichamy V, Arunachalam P, Roopan SM. In vitro Antioxidant and Cytotoxic Capacity of Kappaphycus alvarezii Successive Extracts. Curr Sci. 2020;119(5):790. doi:10.18520/cs/v119/i5/790-798
44. Makkar F, Chakraborty K. First report of dual cyclooxygenase-2 and 5-lipoxygenase inhibitory halogen derivatives from the thallus of intertidal seaweed Kappaphycus alvarezii. Medicinal Chemistry Research. 2018;27(10):2331-2340. doi:10.1007/s00044-018-2239-0
45. Rajaram R, Muralisankar T, Paray BA, Al‐Sadoon MK. Phytochemical profiling and antioxidant capacity of Kappaphycus alvarezii (Doty) Doty collected from seaweed farming sites of tropical coastal environment. Aquac Res. 2021;52(7):3438-3448. doi:10.1111/are.15188
46. Makkar F, Chakraborty K. Unprecedented antioxidative cyclic ether from the red seaweed Kappaphycus alvarezii with anti-cyclooxygenase and lipoxidase activities. Nat Prod Res. 2017;31(10):1131-1141. doi:10.1080/14786419.2016.1230113
47. Sudirman S, Chang HW, Chen CK, Kong ZL. A dietary polysaccharide from Eucheuma cottonii downregulates proinflammatory cytokines and ameliorates osteoarthritis-associated cartilage degradation in obese rats. Food Funct. 2019;10(9):5697-5706. doi:10.1039/C9FO01342C
48. Sudirman S, Hsu YH, He JL, Kong ZL. Dietary polysaccharide-rich extract from Eucheuma cottonii modulates the inflammatory response and suppresses colonic injury on dextran sulfate sodium-induced colitis in mice. PLoS One. 2018;13(10):e0205252. doi:10.1371/journal.pone.0205252
49. Zhang Q, Zhou S, Lim PE, Wei B, Xue C, Xue Y, Tang Q. Kappaphycus Alvarezii Compound Powder Prevents Chemotherapy-Induced Intestinal Mucositis in BALB/c Mice. Nutr Cancer. 2022;74(10):3735-3746. doi:10.1080/01635581.2022.2089699
50. Zhang Q, Yang R, Lim PE, Chin Y, Zhou S, Gao Y, Tang Q. Sun-Dried and Air-Dried Kappaphycus alvarezii Attenuates 5-Fluorouracil-Induced Intestinal Mucositis in Mice. Nutr Cancer. 2022;74(6):2113-2121. doi:10.1080/01635581.2021.1981403
51. James J, Sharma N, Joshi HC, Kumar S, Kumar AP, Hussain A. Extraction and bioactivity assessment of bio‐oil from Kappaphycus alvarezii biomass using the Bligh, Dyer and Soxhlet method. J Am Oil Chem Soc. 2025;102(4):811-822. doi:10.1002/aocs.12931
52. Makkar F, Chakraborty K. Antioxidant and anti-inflammatory oxygenated meroterpenoids from the thalli of red seaweed Kappaphycus alvarezii. Medicinal Chemistry Research. 2018;27(8):2016-2026. doi:10.1007/s00044-018-2210-0
53. Abu Bakar N, Anyanji VU, Mustapha NM, Lim SL, Mohamed S. Seaweed (Eucheuma cottonii) reduced inflammation, mucin synthesis, eosinophil infiltration and MMP-9 expressions in asthma-induced rats compared to Loratadine. J Funct Foods. 2015;19:710-722. doi:10.1016/j.jff.2015.10.011
54. Bajury DM, Rawi MH, Sazali IH, Abdullah A, Sarbini SR. Prebiotic evaluation of red seaweed (Kappaphycus alvarezii) using in vitro colon model. Int J Food Sci Nutr. 2017;68(7):821-828. doi:10.1080/09637486.2017.1309522
55. Sharifuddin Y, Chin YX, Lim PE, Phang SM. Potential Bioactive Compounds from Seaweed for Diabetes Management. Mar Drugs. 2015;13(8):5447-5491. doi:10.3390/md13085447
56. Gong L, Feng D, Wang T, Ren Y, Liu Y, Wang J. Inhibitors of α‐amylase and α‐glucosidase: Potential linkage for whole cereal foods on prevention of hyperglycemia. Food Sci Nutr. 2020;8(12):6320-6337. doi:10.1002/fsn3.1987
57. Lam TP, Tran NVN, Pham LHD, Lai NVT, Dang BTN, Truong NLN, Nguyen-Vo SK, Hoang TL, Mai TT, Tran TD. Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies. Nat Prod Bioprospect. 2024;14(1):4. doi:10.1007/s13659-023-00424-w
58. Lin SR, Chang CH, Tsai MJ, Cheng H, Chen JC, Leong MK, Weng CF. The perceptions of natural compounds against dipeptidyl peptidase 4 in diabetes: from in silico to in vivo. Ther Adv Chronic Dis. 2019;10. doi:10.1177/2040622319875305
59. Fatima MT, Bhat AA, Nisar S, Fakhro KA, Al-Shabeeb Akil AS. The role of dietary antioxidants in type 2 diabetes and neurodegenerative disorders: An assessment of the benefit profile. Heliyon. 2023;9(1):e12698. doi:10.1016/j.heliyon.2022.e12698
60. Caturano A, D’Angelo M, Mormone A, Russo V, Mollica MP, Salvatore T, Galiero R, Rinaldi L, Vetrano E, Marfella R, Monda M, Giordano A, Sasso FC. Oxidative Stress in Type 2 Diabetes: Impacts from Pathogenesis to Lifestyle Modifications. Curr Issues Mol Biol. 2023;45(8):6651-6666. doi:10.3390/cimb45080420
61. Dilworth L, Stennett D, Facey A, Omoruyi F, Mohansingh S, Omoruyi FO. Diabetes and the associated complications: The role of antioxidants in diabetes therapy and care. Biomedicine & Pharmacotherapy. 2024;181:117641. doi:10.1016/j.biopha.2024.117641
62. Yuan T, Yang T, Chen H, Fu D, Hu Y, Wang J, Yuan Q, Yu H, Xu W, Xie X. New insights into oxidative stress and inflammation during diabetes mellitus-accelerated atherosclerosis. Redox Biol. 2019;20:247-260. doi:10.1016/j.redox.2018.09.025
63. Chen X, Xie N, Feng L, Huang Y, Wu Y, Zhu H, Tang J, Zhang Y. Oxidative stress in diabetes mellitus and its complications: From pathophysiology to therapeutic strategies. Chin Med J (Engl). 2025;138(1):15-27. doi:10.1097/CM9.0000000000003230
64. Li X, Xue Y, Zhang Y, Wang Q, Qiu J, Zhang J, Yang C, Zhao Y, Zhang Y. Association between dietary antioxidant capacity and type 2 diabetes mellitus in Chinese adults: a population-based cross-sectional study. Nutr Metab (Lond). 2024;21(1):16. doi:10.1186/s12986-024-00786-z
65. He J yi, Hong Q, Chen B xia, Cui S yuan, Liu R, Cai G yan, Guo J, Chen X mei. Ginsenoside Rb1 alleviates diabetic kidney podocyte injury by inhibiting aldose reductase activity. Acta Pharmacol Sin. 2022;43(2):342-353. doi:10.1038/s41401-021-00788-0
66. Li D, Zhong J, Zhang Q, Zhang J. Effects of anti-inflammatory therapies on glycemic control in type 2 diabetes mellitus. Front Immunol. 2023;14. doi:10.3389/fimmu.2023.1125116
67. Sun Q, Li J, Gao F. New insights into insulin: The anti-inflammatory effect and its clinical relevance. World J Diabetes. 2014;5(2):89-96. doi:10.4239/wjd.v5.i2.89
68. Rai U, Senapati D, Arora MK. Insights on the role of anti-inflammatory and immunosuppressive agents in the amelioration of diabetes. Diabetol Int. 2023;14(2):134-144. doi:10.1007/s13340-022-00607-9
69. Borsani B, De Santis R, Perico V, Penagini F, Pendezza E, Dilillo D, Bosetti A, Zuccotti GV, D’Auria E. The Role of Carrageenan in Inflammatory Bowel Diseases and Allergic Reactions: Where Do We Stand? Nutrients. 2021;13(10). doi:10.3390/nu13103402
70. Komisarska P, Pinyosinwat A, Saleem M, Szczuko M. Carrageenan as a Potential Factor of Inflammatory Bowel Diseases. Nutrients. 2024;16(9). doi:10.3390/nu16091367
71. Al-Jameel SS. Association of diabetes and microbiota: An update. Saudi J Biol Sci. 2021;28(8):4446-4454. doi:10.1016/j.sjbs.2021.04.041
72. Sadagopan A, Mahmoud A, Begg M, Tarhuni M, Fotso M, Gonzalez NA, Sanivarapu RR, Osman U, Latha Kumar A, Mohammed L. Understanding the Role of the Gut Microbiome in Diabetes and Therapeutics Targeting Leaky Gut: A Systematic Review. Cureus. Published online July 8, 2023. doi:10.7759/cureus.41559
73. Li WZ, Stirling K, Yang JJ, Zhang L. Gut microbiota and diabetes: From correlation to causality and mechanism. World J Diabetes. 2020;11(7):293-308. doi:10.4239/wjd.v11.i7.293
74. Xi Y, Xu PF. Diabetes and gut microbiota. World J Diabetes. 2021;12(10):1693-1703. doi:10.4239/wjd.v12.i10.1693
75. Almugadam BS, Liu Y, Chen S min, Wang C hao, Shao C yi, Ren B wei, Tang L, Hatziagelaki E. Alterations of Gut Microbiota in Type 2 Diabetes Individuals and the Confounding Effect of Antidiabetic Agents. J Diabetes Res. 2020;2020:1-14. doi:10.1155/2020/7253978
76. Holscher HD. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes. 2017;8(2):172-184. doi:10.1080/19490976.2017.1290756
77. Rezende ESV, Lima GC, Naves MMV. Dietary fibers as beneficial microbiota modulators: A proposed classification by prebiotic categories. Nutrition. 2021;89:111217. doi:10.1016/j.nut.2021.111217
78. Ojo O, Wang X, Ojo OO, Brooke J, Jiang Y, Dong Q, Thompson T. The Effect of Prebiotics and Oral Anti-Diabetic Agents on Gut Microbiome in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis of Randomised Controlled Trials. Nutrients. 2022;14(23):5139. doi:10.3390/nu14235139
Downloads
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 I Made Agus Mahardika, Putu Oka Samirana

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.





















