A Literature Review and Bibliometric Analysis of Multidrug Therapy (MDT) Resistance in Mycobacterium leprae
DOI:
https://doi.org/10.36733/medicamento.v12i1.12608Keywords:
Bibliometric Analysis, Leprosy, Literature Review, multidrug therapy, Mycobacterium lepraeAbstract
Background: Leprosy remains a global public health problem, and the emergence of resistance to Multidrug Therapy (MDT) poses a serious obstacle to eradication efforts.
Objective: This study aims to systematically review the mechanisms of resistance, the types of drugs involved, and the diagnostic approaches, while mapping research trends through bibliometric analysis.
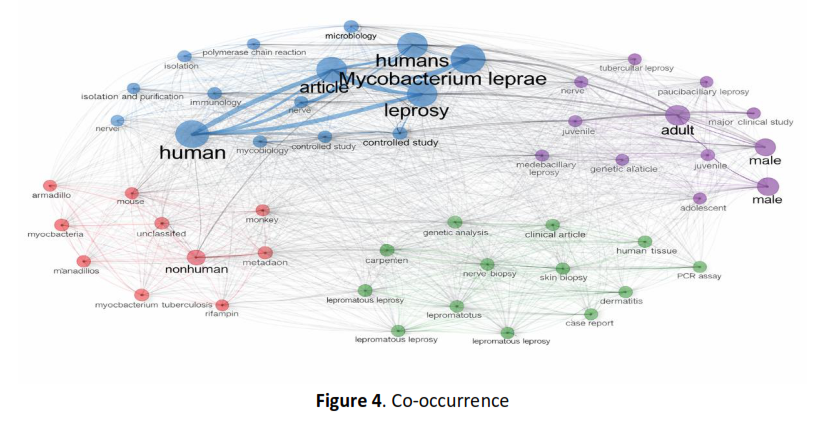
Methods: The study was conducted using a Systematic Literature Review (SLR) method based on PRISMA guidelines with data sourced from Scopus for the 2015–2025 publication period. Of the 11,166 identified articles, only 16 met the inclusion criteria. Furthermore, a bibliometric analysis using the Bibliometrix R package on 2,214 publications was conducted to describe country scientific production, country production over time, co-occurrence, and word cloud.
Results: The results showed that gene mutations rpoB, folP1, and gyrAis a major determinant of resistance to rifampin, dapsone, and ofloxacin, while new mechanisms such as partial duplication folP1 and compensatory mutations in rpoC indicates increasingly complex resistance patterns. India and Brazil contribute the most publications, in line with their high disease burden, while Indonesia's figures are relatively low despite being a major endemic country.
Conclusion: This study emphasizes that MDT resistance requires more precise strategies, including strengthening molecular surveillance, genetic-based diagnostics, and developing locally tailored therapeutic policies. Increased research capacity and international collaboration are also needed to accelerate the achievement of global leprosy elimination targets.
References
1. Sczmanski ADS, Pazin L, Sakae TM, Mazon J. Analysis of the clinical and epidemiological profile of leprosy in Brazil and major regions. Anais Brasileiros de Dermatologia. 2025;100(4):501127. doi:10.1016/j.abd.2025.501127
2. Chen KH, Lin CY, Su SB, Chen KT. Leprosy: A Review of Epidemiology, Clinical Diagnosis, and Management. Wang R, ed. Journal of Tropical Medicine. 2022;2022:1-13. doi:10.1155/2022/8652062
3. Muhaba ES, Geneti SA, Melka D, Mohammed Abdu S. Prevalence, patterns and determinants of peripheral neuropathy among leprosy patients in Northeast Ethiopia: A retrospective study. Blacksell SD, ed. PLoS Negl Trop Dis. 2025;19(3):e0012944. doi:10.1371/journal.pntd.0012944
4. Makhakhe L. Leprosy review. South African Family Practice. 2021;63(1). doi:10.4102/safp.v63i1.5311
5. Tuturop KL, Adimuntja NP, Hutasoit K. FAKTOR RISIKO KEJADIAN PENYAKIT KUSTA DI WILAYAH KERJA PUSKESMAS WAENA KOTA JAYAPURA. JJHSR. 2023;5(2):439-452. doi:10.35971/jjhsr.v5i2.17530
6. Singh I, Sengupta U. Drug Resistance in Mycobacterium Leprae in the Context of Zero Leprosy. Indian Dermatology Online Journal. 2021;12(6):791-795. doi:10.4103/idoj.idoj_599_21
7. Li X, Li G, Yang J, Jin G, Shao Y, Li Y, Wei P, Zhang L. Drug Resistance (Dapsone, Rifampicin, Ofloxacin) and Resistance-Related Gene Mutation Features in Leprosy Patients: A Systematic Review and Meta-Analysis. IJMS. 2022;23(20):12443. doi:10.3390/ijms232012443
8. Verbenko DA, Solomka VS, Kozlova IV, Kubanov AA. The genetic determinants of Mycobacterium leprae resistance to antimicrobial drugs. Vestnik dermatologii i venerologii. 2021;97(6):54-62. doi:10.25208/vdv1292
9. Aubry A, Sammarco Rosa P, Chauffour A, Fletcher ML, Cambau E, Avanzi C. Drug resistance in leprosy: An update following 70 years of chemotherapy. Infectious Diseases Now. 2022;52(5):243-251. doi:10.1016/j.idnow.2022.04.001
10. de Oliveira Ferreira C, Leturiondo A, Santos C, da Silva J, de Andrade Souza M, Rego C, de Souza G, Pinheiro T, Melo G, Rosa P, Mira M, Avanzi C, Talhari C. Classic and new candidate markers for drug resistance in a large cohort of leprosy patients from the Amazon state, Brazil. Doernberg S, ed. Antimicrob Agents Chemother. Published online May 20, 2025:e01550-24. doi:10.1128/aac.01550-24
11. Acebrón-García-de-Eulate M, Blundell TL, Vedithi SC. Strategies for drug target identification in Mycobacterium leprae. Drug Discovery Today. 2021;26(7):1569-1573. doi:10.1016/j.drudis.2021.03.026
12. Ansari A, Kumar R, Ray SK, Patel A, Dwivedi P, Ghosh A, Machado E, Suffys PN, Singh P. Genetic variability, genotyping, and genomics of Mycobacterium leprae. FEMS Microbiology Reviews. 2025;49:fuaf012. doi:10.1093/femsre/fuaf012
13. Silva MJA, Silva CS, Brasil TP, Alves AK, Santos EC, Frota C, Lima KVB, Costa Lima LNG. An update on leprosy immunopathogenesis: systematic review. Front Immunol. 2024;15:1416177. doi:10.3389/fimmu.2024.1416177
14. Sivapatham S, Selvaraj L. Currently available molecular analyses for personalized tumor therapy (Review). Biomed Rep. 2022;17(6):95. doi:10.3892/br.2022.1578
15. Rhodes CJ, Sweatt AJ, Maron BA. Harnessing Big Data to Advance Treatment and Understanding of Pulmonary Hypertension. Circulation Research. 2022;130(9):1423-1444. doi:10.1161/CIRCRESAHA.121.319969
16. Chauffour A, Lecorche E, Reibel F, Mougari F, Raskine L, Aubry A, Jarlier V, Cambau E. Prospective study on antimicrobial resistance in leprosy cases diagnosed in France from 2001 to 2015. Clinical Microbiology and Infection. 2018;24(11):1213.e5-1213.e8. doi:10.1016/j.cmi.2018.06.004
17. Maladan Y, Krismawati H, Wahyuni T, Lorensi Hutapea HM, Rokhmad MF, Parikesit AA. Molecular Docking Analysis of the T450A Mutation of the Gene rpoB Mycobacterium leprae from Leprosy Patients in Papua, West Papua and North Maluku, Indonesia. RJPT. Published online July 19, 2021:3578-3584. doi:10.52711/0974-360X.2021.00619
18. Andrade ESN, Brandão JG, Silva JS da, Coriolano CRF, Rosa PS, Moraes MO, Ferreira C de O, Gomes CM, Araújo WN de. Erratum for Andrade et al., “Antimicrobial Resistance among Leprosy Patients in Brazil: Real-World Data Based on the National Surveillance Plan.” Antimicrob Agents Chemother. 2023;67(7):e00658-23. doi:10.1128/aac.00658-23
19. Chen X, He J, Liu J, You Y, Yuan L, Wen Y. Nested PCR and the TaqMan SNP Genotyping Assay enhanced the sensitivity of drug resistance testing of Mycobacterium leprae using clinical specimens of leprosy patients. Yang R, ed. PLoS Negl Trop Dis. 2019;13(12):e0007946. doi:10.1371/journal.pntd.0007946
20. Chhabra S, Narang T, Sahu S, Sharma K, Shilpa S, Sharma A, Jain S, Singh I, Yadav R, Kaur M, Sharma R, Nadeem M, Pandey P, Minz RW, Dogra S. High frequency of ofloxacin resistance patterns of Mycobacterium leprae from India: An indication to revisit second line anti-leprosy treatment regimen. Journal of Global Antimicrobial Resistance. 2023;35:262-267. doi:10.1016/j.jgar.2023.10.006
21. Mahajan NP, Lavania M, Singh I, Nashi S, Preethish-Kumar V, Vengalil S, Polavarapu K, Pradeep-Chandra-Reddy C, Keerthipriya M, Mahadevan A, Yasha TC, Nandeesh BN, Gnanakumar K, Parry GJ, Sengupta U, Nalini A. Evidence for Mycobacterium leprae Drug Resistance in a Large Cohort of Leprous Neuropathy Patients from India. The American Journal of Tropical Medicine and Hygiene. 2020;102(3):547-552. doi:10.4269/ajtmh.19-0390
22. Kamat D, Narang T, Ahuja M, Lavania M, Dogra S. Case Report: Multidrug-Resistant Mycobacterium leprae in a Case of Smear-Negative Relapse. The American Journal of Tropical Medicine and Hygiene. 2020;102(4):724-727. doi:10.4269/ajtmh.19-0905
23. Benjak A, Avanzi C, Singh P, Loiseau C, Girma S, Busso P, Fontes ANB, Miyamoto Y, Namisato M, Bobosha K, Salgado CG, Da Silva MB, Bouth RC, Frade MAC, Filho FB, Barreto JG, Nery JAC, Bührer-Sékula S, Lupien A, Al-Samie AR, Al-Qubati Y, Alkubati AS, Bretzel G, Vera-Cabrera L, Sakho F, Johnson CR, Kodio M, Fomba A, Sow SO, Gado M, Konaté O, Stefani MMA, Penna GO, Suffys PN, Sarno EN, Moraes MO, Rosa PS, Baptista IMFDi, Spencer JS, Aseffa A, Matsuoka M, Kai M, Cole ST. Phylogenomics and antimicrobial resistance of the leprosy bacillus Mycobacterium leprae. Nat Commun. 2018;9(1):352. doi:10.1038/s41467-017-02576-z
24. Jouet A, Braet SM, Gaudin C, Bisch G, Vasconcellos S, Epaminondas Nicacio de Oliveira do Livramento RE, Prado Palacios YY, Fontes AB, Lucena N, Rosa P, Moraes M, La K, Badalato N, Lenoir E, Ferré A, Clément M, Hasker E, Grillone SH, Abdou W, Said A, Assoumani Y, Attoumani N, Laurent Y, Cambau E, de Jong BC, Suffys PN, Supply P. Hi-plex deep amplicon sequencing for identification, high-resolution genotyping and multidrug resistance prediction of Mycobacterium leprae directly from patient biopsies by using Deeplex Myc-Lep. eBioMedicine. 2023;93:104649. doi:10.1016/j.ebiom.2023.104649
25. Lavania M, Singh I, Turankar RP, Gupta A, Ahuja M, Pathak V, Sengupta U. Enriched whole genome sequencing identified compensatory mutations in the RNA polymerase gene of rifampicin-resistant Mycobacterium leprae strains. IDR. 2018;Volume 11:169-175. doi:10.2147/IDR.S152082
26. Cambau E, Saunderson P, Matsuoka M, Cole ST, Kai M, Suffys P, Rosa PS, Williams D, Gupta UD, Lavania M, Cardona-Castro N, Miyamoto Y, Hagge D, Srikantam A, Hongseng W, Indropo A, Vissa V, Johnson RC, Cauchoix B, Pannikar VK, Cooreman E a. WD, Pemmaraju VRR, Gillini L, leprosy WHO surveillance network of antimicrobial resistance in. Antimicrobial resistance in leprosy: results of the first prospective open survey conducted by a WHO surveillance network for the period 2009–15. Clinical Microbiology and Infection. 2018;24(12):1305-1310. doi:10.1016/j.cmi.2018.02.022
27. Siskawati Y, Effendi EH, Legiawati L, Menaldi SL. Poor treatment compliance leads to a higher mutation for rifampicin resistance in multibacillary leprosy patients. Med J Indones. 2018;27(4):237-243. doi:10.13181/mji.v27i4.1916
28. Marijke Braet S, Jouet A, Aubry A, Van Dyck-Lippens M, Lenoir E, Assoumani Y, Baco A, Mzembaba A, Cambau E, Vasconcellos SEG, Rigouts L, Suffys PN, Hasker E, Supply P, de Jong BC. Investigating drug resistance of Mycobacterium leprae in the Comoros: an observational deep-sequencing study. The Lancet Microbe. 2022;3(9):e693-e700. doi:10.1016/S2666-5247(22)00117-3
29. Reja AHH, Biswas N, Biswas S, Lavania M, Chaitanya VS, Banerjee S, Patra PSM, Gupta UD, Patra PK, Sengupta U, Bhattacharya B. Report of rpoB mutation in clinically suspected cases of drug resistant leprosy: A study from Eastern India. Indian J Dermatol Venereol Leprol. 2015;81(2):155. doi:10.4103/0378-6323.152185
30. Rosa PS, D’Espindula HRS, Melo ACL, Fontes ANB, Finardi AJ, Belone AFF, Sartori BGC, Pires CAA, Soares CT, Marques FB, Branco FJD, Baptista IMFD, Trino LM, Fachin LR V, Xavier MB, Floriano MC, Ura S, Diório SM, Delanina WFB, Moraes MO, Virmond MCL, Suffys PN, Mira MT. Emergence and Transmission of Drug-/Multidrug-resistant Mycobacterium leprae in a Former Leprosy Colony in the Brazilian Amazon. Clinical Infectious Diseases. 2020;70(10):2054-2061. doi:10.1093/cid/ciz570
Downloads
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Sulthon Nurreza Setyawan, Syafri Musthofa, Isnawati Isnawati

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.





















