Revealing Phytopharmaceutical Insights into Jaras Gambo Extract from Uncaria gambir Roxb. against Breast Cancer Using Network Pharmacology and ADME/Tox Approach
DOI:
https://doi.org/10.36733/medicamento.v12i1.12687Keywords:
Breast cancer, gambo, molecular docking, network pharmacology, phytopharmaceutical, Uncaria gambir Roxb.Abstract
Background: Breast cancer remains a leading cause of mortality in women and demands multi-target strategies that can address pathway redundancy and resistance.
Objective: This study aimed to characterize the bioactive constituents of Jaras Gambo Toman extract and evaluate its potential anti-breast cancer activity through an integrated network pharmacology and molecular docking approach.
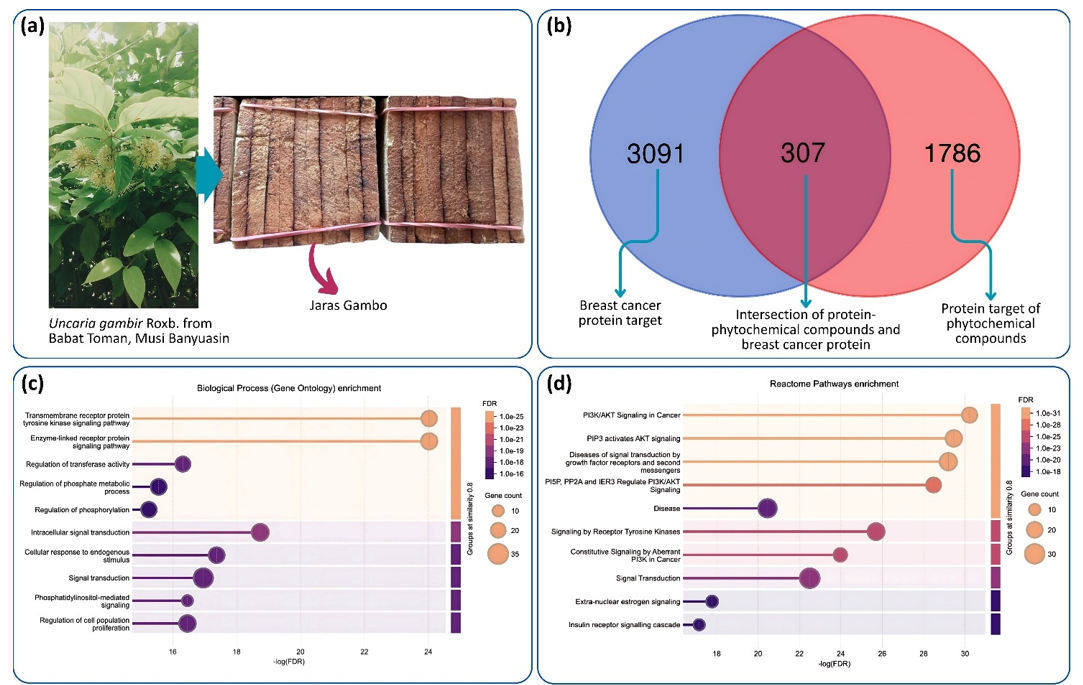
Methods: Jaras Gambo (processed gambier sap mass; Uncaria gambir Roxb.) collected in Babat Toman, South Sumatra, was macerated in ethanol, and the macerate was then filtered and concentrated to obtain a crude extract. Its constituents were profiled by liquid chromatography–high-resolution mass spectrometry (LC–HRMS). Network pharmacology (target prediction, pathway enrichment, and drug-likeness/toxicity screening) was integrated with structure-based molecular docking to prioritize active constituents and predict their interactions with breast cancer–related proteins.
Results: The results of the analysis show therapeutic potential through the significant binding affinity between the bioactive components of the extract and proteins in the regulation of cancer cell proliferation and apoptosis, particularly AKT1, TP53, BCL2, TNF, and EGFR. Molecular interactions are suggested by favorable binding affinity parameters accompanied by the formation of hydrogen and hydrophobic bonds at the active sites of key residues. The involvement of several signaling pathways, such as the PI3K/AKT pathway, p53 signaling pathway, and TNF signaling pathway, may represent key mechanistic pathways of bioactive compounds in targeting disease proteins. Further characterization showed that there are eight main active components, including chlorogenic acid, isoquercitrin, morin hydrate, naringenin, quercetin, eriodictyol, ribofuranoside, and scopoletin, which showed docking profiles comparable to erlotinib across selected targets.
Conclusion: These results suggest the potential of Jaras Gambo Toman extract as a potential source of multi-target bioactive compounds for further breast cancer research.
References
1. Łukasiewicz S, Czeczelewski M, Forma A, Baj J, Sitarz R, Stanisławek A. Breast cancer—epidemiology, risk factors, classification, prognostic markers, and current treatment strategies—An updated review. Cancers (Basel). 2021;13(17). doi:10.3390/cancers13174287
2. Guo L, Kong D, Liu J, et al. Breast cancer heterogeneity and its implication in personalized precision therapy. Exp Hematol Oncol. 2023;12(1). doi:10.1186/s40164-022-00363-1
3. Henry NL, Kim S, Hays RD, et al. Toxicity Index, patient-reported outcomes, and persistence of breast cancer chemotherapy-associated side effects in NRG Oncology/NSABP B-30. NPJ Breast Cancer. 2022;8(1). doi:10.1038/s41523-022-00489-9
4. Pedersen RN, Esen BÖ, Mellemkjær L, et al. The Incidence of breast cancer recurrence 10-32 years after primary diagnosis. J Natl Cancer Inst. 2022;114(3):391-399. doi:10.1093/jnci/djab202
5. Rosso R, D’Alonzo M, Bounous VE, et al. Adherence to adjuvant endocrine therapy in breast cancer patients. Current Oncology. 2023;30(2):1461-1472. doi:10.3390/curroncol30020112
6. Al Khalily IA, Megantara S, Aulifa DL. Targeting molecular pathways in breast cancer using plant-derived bioactive compounds: A comprehensive review. J Exp Pharmacol. 2025;17:375-401. doi:10.2147/JEP.S528132
7. Sohel M, Aktar S, Biswas P, et al. Exploring the anti-cancer potential of dietary phytochemicals for the patients with breast cancer: A comprehensive review. Cancer Med. 2023;12(13):14556-14583. doi:10.1002/cam4.5984
8. Munggari IP, Kurnia D, Deawati Y, Julaeha E. Current research of phytochemical, medicinal and non-medicinal uses of Uncaria gambir Roxb.: A review. Molecules. 2022;27(19). doi:10.3390/molecules27196551
9. Rahmaddiansyah R, Hasani S, Zikrah AA, Arisanty D. The effect of gambier catechin isolate on cervical cancer cell death (hela cell lines). Open Access Maced J Med Sci. 2022;10(B):1293-1297. doi:10.3889/oamjms.2022.8779
10. Wardana AP, Aminah NS, Kristanti AN, et al. Nano Uncaria gambir as chemopreventive agent against breast cancer. Int J Nanomedicine. 2023;18:4471-4484. doi:10.2147/IJN.S403385
11. Gao J, Wang N, Song W, Yuan Y, Teng Y, Liu Z. Mechanisms underlying the synergistic effects of chuanxiong combined with Chishao on treating acute lung injury based on network pharmacology and molecular docking combined with preclinical evaluation. J Ethnopharmacol. 2024;325:117862. doi:https://doi.org/10.1016/j.jep.2024.117862
12. Lee WY, Lee CY, Kim YS, Kim CE. The methodological trends of traditional herbal medicine employing network pharmacology. Biomolecules. 2019;9(8). doi:10.3390/biom9080362
13. Li L, Kar S. Leveraging network pharmacology for drug discovery: Integrative approaches and emerging insights. Med Drug Discov. 2025;27. doi:10.1016/j.medidd.2025.100220
14. Ferreira LLG, Andricopulo AD. ADMET modeling approaches in drug discovery. Drug Discov Today. 2019;24(5):1157-1165. doi:10.1016/j.drudis.2019.03.015
15. Muhammed MT, Aki-Yalcin E. Molecular docking: Principles, advances, and its applications in drug discovery. Lett Drug Des Discov. 2024;21(3):480-495. doi:https://doi.org/10.2174/1570180819666220922103109
16. Adelusi TI, Oyedele AQK, Boyenle ID, et al. Molecular modeling in drug discovery. Inform Med Unlocked. 2022;29. doi:10.1016/j.imu.2022.100880
17. Aries Seka O, Kiagus Ahmad Roni I, Elfidiah I, Author C. Optimizing the effect of gambir sap extract as an organic inhibitor to reduce scale formation at pertamina hulu energi (OK/RT) Peninjauan District, OKU Regency. Int j adv multidisc res stud. 2023;3(5):73-77.
18. Kızıltaş H, Bingol Z, Gören AC, et al. LC-HRMS profiling and antidiabetic, anticholinergic, and antioxidant activities of aerial parts of kınkor (Ferulago stellata). Molecules. 2021;26(9). doi:10.3390/molecules26092469
19. Filimonov DA, Rudik A V., Dmitriev A V., Poroikov V V. Computer-aided estimation of biological activity profiles of drug-like compounds taking into account their metabolism in human body. Int J Mol Sci. 2020;21(20):1-13. doi:10.3390/ijms21207492
20. Nugraha SE, Marianne M, Syahputra RA, et al. Efficacy of Carica papaya leaves extract for treating thrombocytopenia: An in silico and in vivo study in rat model. Adv Anim Vet Sci. 2024;12(7):1325-1334. doi:10.17582/journal.aavs/2024/12.7.1325.1334
21. Astuti PDY, Fadilah F, Promsai S, Bahtiar A. Integrating molecular docking and molecular dynamics simulations to evaluate active compounds of Hibiscus schizopetalus for obesity. J Appl Pharm Sci. 2024;14(4):176-187. doi:10.7324/JAPS.2024.158550
22. Desa S, Osman A, Hyslop R. In silico assessment of drug-like properties of phytocannabinoids in Cannabis sativa. EJSMT. 2017;4(2):1-7. doi:10.37134/ejsmt.vol4.2.1.2017
23. Ramírez D, Caballero J. Is it reliable to take the molecular docking top scoring position as the best solution without considering available structural data? Molecules. 2018;23(5). doi:10.3390/molecules23051038
24. Purnama A, Rizki D, Qanita I, et al. Molecular docking investigation of calotropone as a potential natural therapeutic agent against pancreatic cancer. J Adv Pharm Technol Res. 2022;13(1):44-49. doi:10.4103/japtr.japtr_143_21
25. Slika H, Mansour H, Wehbe N, et al. Therapeutic potential of flavonoids in cancer: ROS-mediated mechanisms. Biomedicine and Pharmacotherapy. 2022;146. doi:10.1016/j.biopha.2021.112442
26. Qi Y. Receptor tyrosine kinases in breast cancer treatment: unraveling the potential. Am J Cancer Res. 2024;14(9):4172-4196. doi:10.62347/kivs3169
27. Pottier C, Fresnais M, Gilon M, Jérusalem G, Longuespée R, Sounni NE. Tyrosine kinase inhibitors in cancer: Breakthrough and challenges of targeted therapy. Cancers (Basel). 2020;12(3). doi:10.3390/cancers12030731
28. Abotaleb M, Liskova A, Kubatka P, Büsselberg D. Therapeutic potential of plant phenolic acids in the treatment of cancer. Biomolecules. 2020;10(2). doi:10.3390/biom10020221
29. Huang D, Yang J, Zhang Q, et al. Design, synthesis, and biological evaluation of 2,4-dimorpholinopyrimidine-5-carbonitrile derivatives as orally bioavailable PI3K inhibitors. Front Pharmacol. 2024;15. doi:10.3389/fphar.2024.1467028
30. Suhail M, AlZahrani WM, Shakil S, et al. Analysis of some flavonoids for inhibitory mechanism against cancer target phosphatidylinositol 3-kinase (PI3K) using computational tool. Front Pharmacol. 2023;14. doi:10.3389/fphar.2023.1236173
31. Silva GB da, Rocha KG, Bagatini MD, Kempka AP. Anticancer properties of phenolic acids and cell death signaling pathways: A 20-year bibliometric analysis (2003–2023). Food Biosci. 2025;63. doi:10.1016/j.fbio.2024.105741
32. Guan L, Yang H, Cai Y, et al. ADMET-score – a comprehensive scoring function for evaluation of chemical drug-likeness. Med Chem Commun. 2019;10(1):148-157. doi:10.1039/C8MD00472B
33. Chander S, Tang CR, Al-Maqtari HM, et al. Synthesis and study of anti-HIV-1 RT activity of 5-benzoyl-4-methyl-1,3,4,5-tetrahydro-2H-1,5-benzodiazepin-2-one derivatives. Bioorg Chem. 2017;72:74-79. doi:10.1016/j.bioorg.2017.03.013
34. Wee P, Wang Z. Epidermal growth factor receptor cell proliferation signaling pathways. Cancers (Basel). 2017;9(5). doi:10.3390/cancers9050052
35. Van Nguyen C, Nguyen QT, Vu HTN, Phung HT, Pham KH, Le RD. Combined p53 and Bcl2 immunophenotypes in prognosis of vietnamese invasive breast carcinoma: A single institutional retrospective analysis. Technol Cancer Res Treat. 2020;19. doi:10.1177/1533033820983081
36. Shi P, Xu J, Cui H. The recent research progress of NF-κB signaling on the proliferation, migration, invasion, immune escape and drug resistance of glioblastoma. Int J Mol Sci. 2023;24(12). doi:10.3390/ijms241210337
37. Qodir N, Pramudhito D, Legiran, et al. Tumor necrosis factor-alpha and its association with breast cancer: A systematic review. World J Oncol. 2025;16(2):143-151. doi:10.14740/wjon2532
38. Gharge S, Gudasi S, Koli R. Targeting MDA-MB-231 cells using Ailanthus excelsa Roxb: A virtual binding evaluation, molecular flexibility analysis, and cytotoxicity study. In Silico Research in Biomedicine. 2025;1:100064. doi:10.1016/j.insi.2025.100064
39. Elsori D, Pandey P, Ramniwas S, et al. Naringenin as potent anticancer phytocompound in breast carcinoma: from mechanistic approach to nanoformulations based therapeutics. Front Pharmacol. 2024;15. doi:10.3389/fphar.2024.1406619
40. Hsu PH, Chen WH, Juanlu C, et al. Hesperidin and chlorogenic acid synergistically inhibit the growth of breast cancer cells via estrogen receptor/mitochondrial pathway. Life. 2021;11(9). doi:10.3390/life11090950
41. Bender O, Atalay A. Evaluation of anti-proliferative and cytotoxic effects of chlorogenic acid on breast cancer cell lines by real-time, label-free and high-throughput screening. Marmara Pharm J. 2018;22(2):173-179. doi:10.12991/mpj.2018.54
42. Ranganathan S, Halagowder D, Sivasithambaram ND. Quercetin suppresses twist to induce apoptosis in MCF-7 breast cancer cells. PLoS One. 2015;10(10). doi:10.1371/journal.pone.0141370
43. Wu ZY, Qiu KY, Gai YJ, Wu JH, Zhou BX, Shi QF. Quercetin: A natural ally in combating breast cancer. Int J Nanomedicine. 2025;20:9155-9177. doi:10.2147/IJN.S518174
44. Hashemzaei M, Far AD, Yari A, et al. Anticancer and apoptosis-inducing effects of quercetin in vitro and in vivo. Oncol Rep. 2017;38(2):819-828. doi:10.3892/or.2017.5766
45. Di Camillo Orfali G, Duarte AC, Bonadio V, et al. Review of anticancer mechanisms of isoquercitin. World J Clin Oncol. 2016;7(2):189-199. doi:10.5306/wjco.v7.i2.189
46. Gao XY, Li XY, Zhang CY, Bai CY. Scopoletin: a review of its pharmacology, pharmacokinetics, and toxicity. Front Pharmacol. 2024;15. doi:10.3389/fphar.2024.1268464
47. Singh SK, Srivastav AK, Chaurasiya S, Patel S, Kumar U, Kulhari H. Nanoencapsulation of morin hydrate with BSA for sustained drug release in colorectal carcinoma cells: experimental and computational approach. Frontiers in Drug Delivery. 2025;5. doi:10.3389/fddev.2025.1623317
Downloads
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Rafiqah Nur Viviani, Nathasya Shasykirana Mahendra, Shaum Shiyan, Galih Pratiwi

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.





















