Understanding the Safety Profile of Imatinib in Asian Chronic Myeloid Leukemia Patients: A Systematic Review
DOI:
https://doi.org/10.36733/medicamento.v12i1.13296Keywords:
adverse drug reactions, Asia, hematologic and non-hematologic toxicity, imatinib mesylate, tyrosine kinase inhibitorAbstract
Background: The treatment of chronic myeloid leukemia has advanced substantially since the introduction of tyrosine kinase inhibitors, particularly imatinib. However, adverse events associated with imatinib may affect adherence and quality of life, highlighting the importance of understanding safety outcomes across populations.
Objective: This systematic review aimed to synthesize evidence on hematologic and non-hematologic adverse events associated with imatinib use among chronic myeloid leukemia patients in Asia.
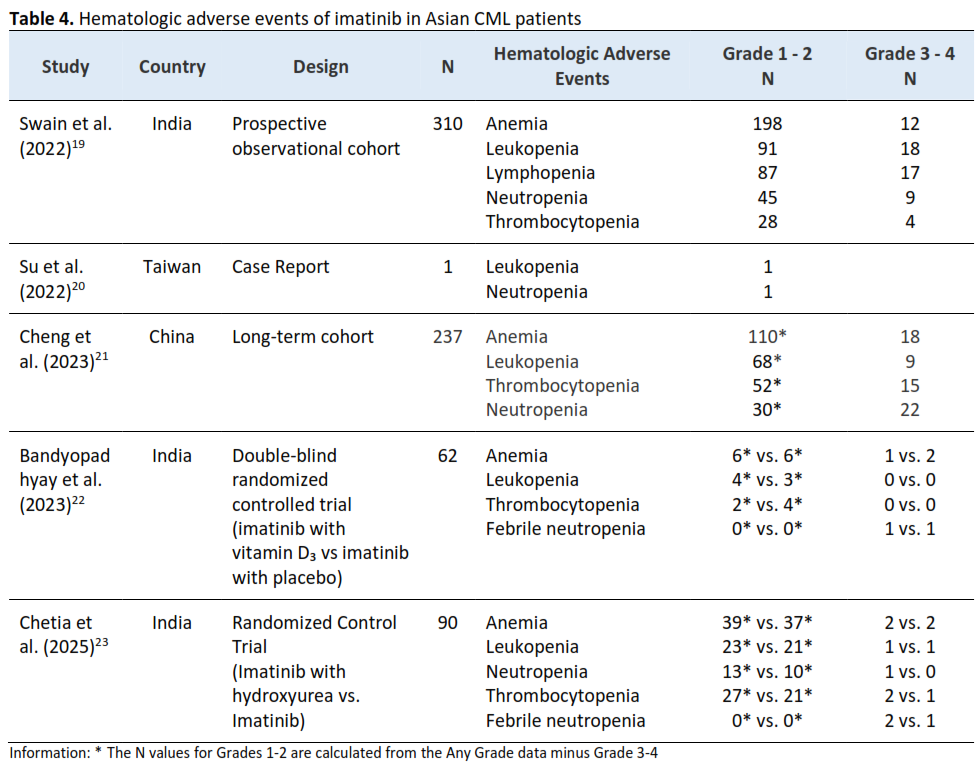
Methods: A systematic literature search was conducted in PubMed, Scopus, and ScienceDirect for studies published between January 2020 and July 2025. Eligible studies were synthesized narratively due to methodological heterogeneity. Adverse events were extracted as reported and graded using standardized toxicity criteria, and causality assessment was applied when available. Study quality was evaluated using established critical appraisal tools. Five studies from India, China, and Taiwan met the inclusion criteria.
Results: The most frequent hematologic adverse event was anemia, followed by neutropenia and thrombocytopenia. Common non-hematologic adverse events included gastrointestinal symptoms, peripheral or periorbital edema, muscle cramps, and hyperpigmentation, with regional variations. Most events were mild to moderate (grades 1–2), while severe fluid retention, including pleural and pericardial effusions, was reported in isolated cases. No studies reported permanent discontinuation of imatinib due to adverse events.
Conclusion: This review summarizes imatinib-related adverse events among chronic myeloid leukemia patients from selected Asian regions—East and South Asia, specifically India, China, and Taiwan—showing predominantly mild to moderate toxicities and providing practice-informed insights for clinical monitoring. However, the absence of data from other Asian regions precludes generalization to the entire Asian continent.
References
1. Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2022 update on diagnosis, therapy, and monitoring. Am J Hematol. 2022;97(9):1236-1256. doi:10.1002/ajh.26642
2. Lee H, Basso IN, Kim DDH. Target spectrum of the BCR-ABL tyrosine kinase inhibitors in chronic myeloid leukemia. Int J Hematol. 2021;113(5):632-641. doi:10.1007/s12185-021-03126-6
3. Hochhaus A, Baccarani M, Silver RT, Schiffer C, Apperley JF, Cervantes F, Clark RE, Cortes JE, Deininger MW, Guilhot F, Hjorth-Hansen H, Hughes TP, Janssen JJWM, Kantarjian HM, Kim DW, Larson RA, Lipton JH, Mahon FX, Mayer J, Nicolini F, Niederwieser D, Pane F, Radich JP, Rea D, Richter J, Rosti G, Rousselot P, Saglio G, Saußele S, Soverini S, Steegmann JL, Turkina A, Zaritskey A, Hehlmann R. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia. 2020;34(4):966-984. doi:10.1038/s41375-020-0776-2
4. Chuah C, Koh LP, Numbenjapon T, Zang DY, Ong KH, Do YR, Ohkura M, Ono C, Viqueira A, Cortes JE, Brümmendorf TH. Efficacy and safety of bosutinib versus imatinib for newly diagnosed chronic myeloid leukemia in the Asian subpopulation of the phase 3 BFORE trial. Int J Hematol. 2021;114(1):65-78. doi:10.1007/s12185-021-03144-4
5. Wu Y, Zhang H, Luo S, Zhang X, Tan M, Chen J, Huang P, Chen Y, Du Z. The Global Burden of Leukemia, 1990–2021: A Systematic Analysis of Prevalence, DALYs, and Risk Factors From the Global Burden of Disease Study. Med Res. Published online November 17, 2025. doi:10.1002/mdr2.70036
6. Daltveit DS, Morgan E, Colombet M, Steliarova-Foucher E, Bendahhou K, Marcos-Gragera R, Rongshou Z, Smith A, Wei H, Soerjomataram I. Global patterns of leukemia by subtype, age, and sex in 185 countries in 2022. Leukemia. 2025;39(2):412-419. doi:10.1038/s41375-024-02452-y
7. Phukan A, Mandal PK, Dolai TK. Efficacy and safety profile of generic imatinib in patients with newly diagnosed chronic myeloid leukemia-chronic phase: sharing experience of a hemato-oncology center from eastern India. Ann Hematol. 2021;100(1):85-96. doi:10.1007/s00277-020-04289-8
8. Cortes J, Pavlovsky C, Saußele S. Chronic myeloid leukaemia. Lancet. 2021;398(10314):1914-1926. doi:10.1016/S0140-6736(21)01204-6
9. Wolfe HR, Rein LAM. The Evolving Landscape of Frontline Therapy in Chronic Phase Chronic Myeloid Leukemia (CML). Curr Hematol Malig Rep. 2021;16(5):448-454. doi:10.1007/s11899-021-00655-z
10. Woudberg R, Sinanovic E. Cost-effectiveness of tyrosine kinase inhibitor treatment strategies for chronic myeloid leukemia in South Africa. Front Pharmacol. 2025;15:1511603. doi:10.3389/fphar.2024.1511603
11. Clark SE, Marcum ZA, Radich J, Etzioni R, Basu A. Temporal effect of imatinib adherence on time to remission in chronic myeloid leukemia patients. J Oncol Pharm Pract. 2024;30(8):1343-1351. doi:10.1177/10781552231212207
12. Efficace F, Cottone F, Yanez B, Kota V, Castagnetti F, Caocci G, Bonifacio M, Patriarca A, Capodanno I, Miggiano MC, Tiribelli M, Breccia M, Luciano L, Giai V, Iurlo A, Abruzzese E, Fava C, Dinner S, Altman JK, Rosti G, Cortes J, Vignetti M, Cella D. Patient‐reported symptom monitoring and adherence to therapy in patients with newly diagnosed chronic myeloid leukemia. Cancer. 2024;130(2):287-299. doi:10.1002/cncr.35021
13. Obeng-Kusi M, MacDonald K, van Lierde MA, Lee CS, De Geest S, Abraham I. No margin for non-adherence: Probabilistic kaplan-meier modeling of imatinib non-adherence and treatment response in CML (ADAGIO study). Leuk Res. 2021;111(October):106734. doi:10.1016/j.leukres.2021.106734
14. Adattini JA, Gross AS, Wong Doo N, McLachlan AJ. Real‐world efficacy and safety outcomes of imatinib treatment in patients with chronic myeloid leukemia: An Australian experience. Pharmacol Res Perspect. 2022;10(5). doi:10.1002/prp2.1005
15. Kyriacou NM, Gross AS, McLachlan AJ. Inter‐Ethnic Differences in the Efficacy and Safety of Tyrosine Kinase Inhibitors Used in Oncology: Insights From Phase 3 Clinical Trials. Clin Transl Sci. 2025;18(5):4-8. doi:10.1111/cts.70224
16. Aslam HM, Shumaila IM, Merchant AA, Muhammad MG, Yasir J, Faizee FA, Qadir MA. Hematologic Response and Frequency of Side Effects in Chronic Myeloid Leukemia Patients Treated with Imatinib (GLIVEC): A South-East Asian Experience. Blood. 2017;130(Suppl 1):5257. doi:10.1182/blood.V130.Suppl_1.5257.5257
17. Ning L, Hu C, Lu P, Que Y, Zhu X, Li D. Trends in disease burden of chronic myeloid leukemia at the global, regional, and national levels: a population-based epidemiologic study. Exp Hematol Oncol. 2020;9(1):29. doi:10.1186/s40164-020-00185-z
18. Aromataris E, Lockwood C, Porritt K, Pilla B, Jordan Z. JBI Manual for Evidence Synthesis. (Aromataris E, Lockwood C, Porritt K, Pilla B, Jordan Z, eds.). JBI; 2024. doi:10.46658/JBIMES-24-01
19. Swain TR, Goutam S, Jena RK, Rout N. Analysis of adverse drug reactions of imatinib in chronic myeloid leukemia patients. Indian J Pharmacol. 2022;54(5):349-352. doi:10.4103/ijp.ijp_844_21
20. Su PY, Tan BF, Fu CM, Chen CN, Chou AK, Kung PJ, Liao LC, Li MJ. Concurrence of imatinib-induced massive pleural/pericardial effusion and Campylobacter bacteremia in an adolescent with chronic myeloid leukemia. J Infect Chemother. 2022;28(1):103-107. doi:10.1016/j.jiac.2021.10.002
21. Cheng F, Yuan G, Li Q, Cui Z, Li W. Long-term outcomes of frontline imatinib therapy for chronic myeloid leukemia in China. Front Oncol. 2023;13:1172910. doi:10.3389/fonc.2023.1172910
22. Bandyopadhyay A, Palepu S, Dhamija P, Nath UK, Chetia R, Bakliwal A, Vaniyath S, Chattopadhyay D, Handu S. Safety and efficacy of Vitamin D 3 supplementation with Imatinib in Chronic Phase- Chronic Myeloid Leukaemia: an Exploratory Randomized Controlled Trial. BMJ Open. 2023;13(8):e066361. doi:10.1136/bmjopen-2022-066361
23. Chetia R, Palepu S, Dutta V, Bandyopadhyay A, Mathew A, Vaniyath S, Bakliwal A, Chattopadhyay D, Rajoreya A, Dhamija P, Naithani M, Singh N, Nath UK. Upfront Combined Hydroxyurea and Imatinib versus Imatinib Monotherapy in Newly Diagnosed Chronic Phase Chronic Myeloid Leukemia: A Randomized Controlled Trial. South Asian J Cancer. 2025;14(03):608-614. doi:10.1055/s-0044-1789579
24. Tangkitchot J, Tantiworawit A, Niprapan P, Wongsarikan N, Srichairatanakool S, Punnachet T, Hantrakun N, Piriyakhuntorn P, Rattanathammethee T, Chai-Adisaksopha C, Rattarittamrong E, Norasetthada L, Hantrakool S. Comparative Clinical Outcomes and Safety of Generic Versus Original Imatinib in the Treatment of Chronic Myeloid Leukemia: A Real-World Cohort Study from Thailand. J Clin Med. 2025;14(11):3695. doi:10.3390/jcm14113695
25. Talapphet N, Kim MM. Imatinib mesylate promotes melanogenesis through the modulation of p38 and MITF in murine cells. Toxicol Res. 2025;41(1):61-70. doi:10.1007/s43188-024-00267-8
26. Khan L, Salsabil MA, Ferdous J, Haque MR. Evaluation of Myelosupression during Imatinib Mesylate Therapy in Patients with Chronic Myeloid Leukaemia. J Armed Forces Med Coll Bangladesh. 2020;15(2):189-192. doi:10.3329/jafmc.v15i2.50833
27. McLigeyo A, Rajab J, Ezzi M, Oyiro P, Bett Y, Odhiambo A, Ong’ondi M, Mwanzi S, Gatua M, Abinya N. Cytopenia among CML Patients on Imatinib in Kenya: Types, Grades, and Time Course. Adv Hematol. 2020;2020:1-5. doi:10.1155/2020/7696204
28. Hochhaus A, Larson RA, Guilhot F, Radich JP, Branford S, Hughes TP, Baccarani M, Deininger MW, Cervantes F, Fujihara S, Ortmann CE, Menssen HD, Kantarjian H, O’Brien SG, Druker BJ. Long-Term Outcomes of Imatinib Treatment for Chronic Myeloid Leukemia. N Engl J Med. 2017;376(10):917-927. doi:10.1056/NEJMoa1609324
29. Jabbour E, Kantarjian H. Chronic myeloid leukemia: 2025 update on diagnosis, therapy, and monitoring. Am J Hematol. 2024;99(11):2191-2212. doi:10.1002/ajh.27443
30. Adiwidjaja J, Gross AS, Boddy A V., McLachlan AJ. Physiologically‐based pharmacokinetic model predictions of inter‐ethnic differences in imatinib pharmacokinetics and dosing regimens. Br J Clin Pharmacol. 2022;88(4):1735-1750. doi:10.1111/bcp.15084
31. Xia T, Shu B, Peng Y, Wang B. Post-marketing safety signals of imatinib: pharmacovigilance insights from the FDA Adverse Event Reporting System (FAERS) and implications for clinical practice. Eur J Clin Pharmacol. 2025;81(9):1343-1353. doi:10.1007/s00228-025-03872-0
Downloads
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Niky Budiarti, Zullies Ikawati, Arief Nurrochmad, Thendi Abdul Arief

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.





















