Biochemoinformatics Screening of Chemical Constituents from Laportea aestuans L. Leaves as Potential Prostate Cancer Inhibitors
DOI:
https://doi.org/10.36733/medicamento.v12i1.12565Keywords:
ADME profiling, CDK2, Laportea aestuans L., molecular docking, natural compounds, prostate cancerAbstract
Background: Prostate cancer remains a major cause of morbidity among men worldwide, with current therapies limited by resistance and toxicity. Natural compounds offer promising alternatives due to their structural diversity and biological compatibility.
Objective: This study evaluated seven bioactive constituents from Laportea aestuans L. leaves as potential inhibitors of prostate cancer-related targets using molecular docking and pharmacokinetic profiling.
Methods: Seven bioactive compounds were identified through phytochemical literature review. Molecular docking simulations were executed using AutoDock4, with interaction analysis validated via PyMOL and UCSF Chimera. Furthermore, pharmacokinetic properties and toxicological profiles were predicted using the ADMETlab webserver to assess drug-likeness and safety parameters.
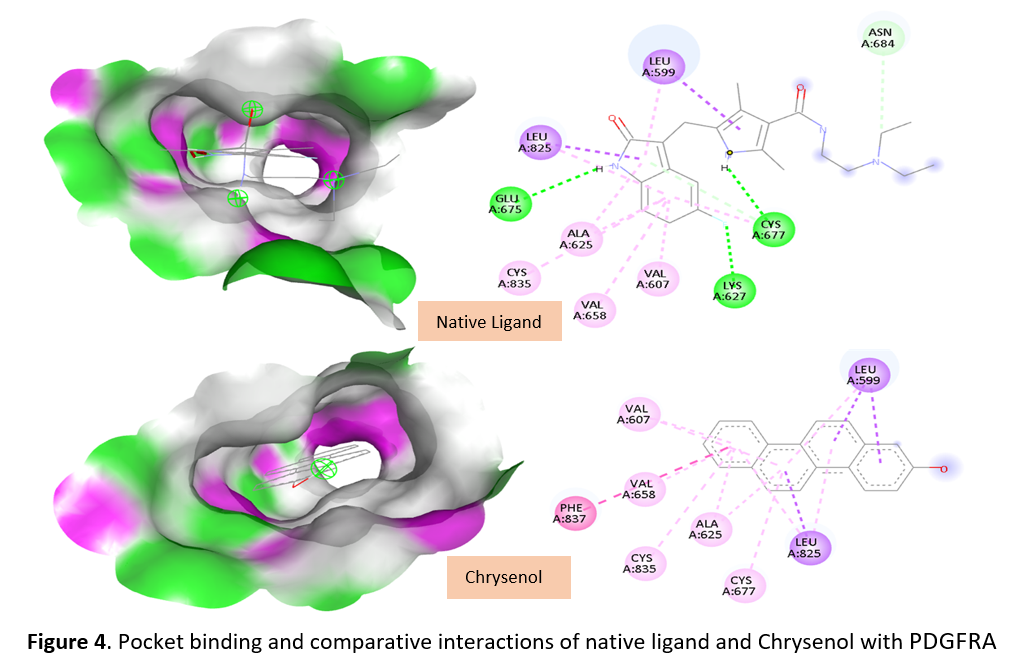
Results: Docking simulations revealed that LA6 exhibited the strongest affinity toward CDK2 (–9.97 kcal/mol) through hydrogen bonding with Asp145 and Lys33, while Chrysenol bound PDGFRA (–9.12 kcal/mol) at Glu644 and Val658. Compounds LA2, LA4, and LA5 also showed stable interactions with GSK-3β, suggesting modulation of Wnt signaling. ADME analysis indicated high gastrointestinal absorption and compliance with Lipinski’s Rule of Five, whereas LA6 and Chrysenol demonstrated mutagenicity and hepatotoxicity risks. Distribution profiling revealed high plasma protein binding (>90%) and moderate blood-brain barrier permeability, particularly for LA6.
Conclusion: These findings highlight L. aestuans L. derivatives as promising lead candidates for prostate cancer therapy, though further in vitro and in vivo validation is required to confirm efficacy and optimize safety. This integrative computational approach underscores the role of bioinformatics in accelerating natural product-based drug discovery.
References
1. National Cancer Institute. Understanding Cancer [Internet]. 2015 [cited 2020 Oct 10]. Available from: https://www.cancer.gov/about-cancer/understanding/what-is-cancer
2. End Cancer As We Know It. Accessed April 28, 2026. https://www.cancer.org
3. Khan GJ, Khan MA, Khan T, et al. Pharmacological effects and potential therapeutic targets of DT-13. Biomed Pharmacother. 2018;97:255–63. doi:10.1016/j.biopha.2017.10.101
4. Shorning BY, Dass MS, Smalley MJ, Pearson HB. The PI3K-AKT-mTOR Pathway and Prostate Cancer: At the Crossroads of AR, MAPK, and WNT Signaling. Int J Mol Sci. 2020;21(12):4507. doi:10.3390/ijms21124507
5. Hare SH, Harvey AJ. mTOR function and therapeutic targeting in breast cancer. Am J Cancer Res. 2017;7(3):383-404.
6. Aladesanmi AJ, Famuyiwa FG, Oriola AO, Oguntimehin SA, Aiyedun PO, Arthur G. Cytotoxic Activity of Selected Nigerian Medicinal Plants. J Herbs Spices Med Plants. 2020;26(2):203-217. doi:10.1080/10496475.2019.1706214
7. Prostate Cancer. CancerQuest. Accessed April 29, 2026. https://cancerquest.org/patients/cancer-type/prostate-cancer
8. USDA Plants Database Plant Profile General. Accessed April 28, 2026. https://plants.sc.egov.usda.gov/plant-profile/LAAE
9. Sc O, Cs C, Nwaokezi C. Antioxidant enzyme activities and chemopreventive potentials of Laportea aestuans on urinary inflammatory markers using albino rats. Published online January 1, 2017:288-291.
10. Cao Z, Kyprianou N. Mechanisms navigating the TGF-β pathway in prostate cancer. Asian J Urol. 2015;2(1):11-18. doi:10.1016/j.ajur.2015.04.011
11. Liu Z, Guo F, Wang Y, et al. BATMAN-TCM: a Bioinformatics Analysis Tool for Molecular mechANism of Traditional Chinese Medicine. Sci Rep. 2016;6(1):21146. doi:10.1038/srep21146
12. Xie F, Ding X, Zhang QY. An update on the role of intestinal cytochrome P450 enzymes in drug disposition. Acta Pharm Sin B. 2016;6(5):374-383. doi:10.1016/j.apsb.2016.07.012
13. Zhang X, Zhou H, Su Y. Targeting truncated RXRα for cancer therapy. Acta Biochim Biophys Sin. 2016;48(1):49-59. doi:10.1093/abbs/gmv104
14. Naimi A, Soltan M, Amjadi E, Goli P, Kefayat A. Androgen Receptor Expression and Its Correlation with Clinicopathological Parameters in Iranian Patients with Triple Negative Breast Cancer. Iran J Pathol. 2020;15(3):239-244. doi:10.30699/ijp.2020.112819.2224
15. Goldman J, Eckhardt SG, Borad MJ, et al. Phase I dose-escalation trial of the oral investigational Hedgehog signaling pathway inhibitor TAK-441 in patients with advanced solid tumors. Clin Cancer Res Off J Am Assoc Cancer Res. 2015;21(5):1002-1009. doi:10.1158/1078-0432.CCR-14-1234
16. Dietrich C, Trub A, Ahn A, et al. INX-315, a Selective CDK2 Inhibitor, Induces Cell Cycle Arrest and Senescence in Solid Tumors. Cancer Discov. 2024;14(3):446-467. doi:10.1158/2159-8290.CD-23-0954
17. Knudsen ES, Witkiewicz AK, Sanidas I, Rubin SM. Targeting CDK2 for cancer therapy. Cell Rep. 2025;44(8):116140. doi:10.1016/j.celrep.2025.116140
18. Liang J, Pan Y, Yang J, Zeng D, Li J. WNT signaling in cancer: molecular mechanisms and potential therapies. Mol Biomed. 2025;6:83. doi:10.1186/s43556-025-00327-x
19. Reddy SK, Reddy SVG, Basha SH. Discovery of novel PDGFR inhibitors targeting non-small cell lung cancer using a multistep machine learning assisted hybrid virtual screening approach. RSC Adv. 2025;15(2):851-869. doi:10.1039/D4RA06975G
20. El-Hachem N, Haibe-Kains B, Khalil A, Kobeissy FH, Nemer G. AutoDock and AutoDockTools for Protein-Ligand Docking: Beta-Site Amyloid Precursor Protein Cleaving Enzyme 1(BACE1) as a Case Study. Methods Mol Biol. 2017;1598:391-403. doi:10.1007/978-1-4939-6952-4_20
21. Forli S, Huey R, Pique ME, Sanner MF, Goodsell DS, Olson AJ. Computational protein-ligand docking and virtual drug screening with the AutoDock suite. Nat Protoc. 2016;11(5):905-919. doi:10.1038/nprot.2016.051
22. (PDF) SAWAR OTAK. ResearchGate. Published online January 5, 2026. doi:10.29342/cnj.v2i1.54
23. Persidsky Y, Ramirez SH, Haorah J, Kanmogne GD. Blood-brain barrier: structural components and function under physiologic and pathologic conditions. J Neuroimmune Pharmacol Off J Soc NeuroImmune Pharmacol. 2006;1(3):223-236. doi:10.1007/s11481-006-9025-3
24. Acikgoz E, Ozdil B, Oktem G, Aktug H, Ragbetli MC, Tas C. GSK-3 inhibitor induces apoptosis and cell cycle arrest in CD133+/CD44+ prostate cancer cells through modulation of Notch signaling pathway. Mol Biol Rep. 2025;52(1):892. doi:10.1007/s11033-025-10977-3
25. Hua L, Anjum F, Shafie A, et al. Identifying promising GSK3β inhibitors for cancer management: a computational pipeline combining virtual screening and molecular dynamics simulations. Front Chem. 2023;11. doi:10.3389/fchem.2023.1200490
26. Siskin M, Economides MP, Wise DR. Cyclin-Dependent Kinase Inhibition in Prostate Cancer: Past, Present, and Future. Cancers. 2025;17(5):774. doi:10.3390/cancers17050774
27. Shiota M, Fujimoto N, Kashiwagi E, Eto M. The Role of Nuclear Receptors in Prostate Cancer. Cells. 2019;8(6):602. doi:10.3390/cells8060602
28. Mayr L, Neyazi S, Schwark K, et al. Effective targeting of PDGFRA-altered high-grade glioma with avapritinib. Cancer Cell. 2025;43(4):740-756.e8. doi:10.1016/j.ccell.2025.02.018
29. Zhang R, Yao Y, Gao H, Hu X. Mechanisms of angiogenesis in tumour. Front Oncol. 2024;14. doi:10.3389/fonc.2024.1359069
30. Zheng Y, Trivedi T, Lin RC, et al. Loss of the vitamin D receptor in human breast and prostate cancers strongly induces cell apoptosis through downregulation of Wnt/β-catenin signaling. Bone Res. 2017;5(1):17023. doi:10.1038/boneres.2017.23
Downloads
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 I Wayan Surya Rahadi, Rina Herowati

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.





















